What type of IVF is an isotonic solution?
What type of IVF is an isotonic solution?
Normal saline is the isotonic solution of choice for expanding the extracellular fluid (ECF) volume because it does not enter the intracellular fluid (ICF). It is administered to correct extracellular fluid volume deficit because it remains within the ECF.
What is an isotonic IV fluid?
Isotonic solutions are IV fluids that have a similar concentration of dissolved particles as blood. An example of an isotonic IV solution is 0.9% Normal Saline (0.9% NaCl). Isotonic solutions are used for patients with fluid volume deficit (also called hypovolemia) to raise their blood pressure.
Why would you give a patient isotonic solution?
Isotonic Solutions These fluids are useful when the patient has lost fluid volume from blood loss, trauma, or dehydration due to excessive nausea/vomiting or diarrhea.
What do you mean by isotonic solution?
Isotonic solution: A solution that has the same salt concentration as cells and blood. Isotonic solutions are commonly used as intravenously infused fluids in hospitalized patients.
Why is isotonic saline used for dehydration?
The simplest approach is to replace dehydration losses with 0.9% saline. This ensures that the administered fluid remains in the extracellular (intravascular) compartment, where it will do the most good to support blood pressure and peripheral perfusion.
Why is isotonic fluid used to treat dehydration?
Isotonic IV solutions restore fluid volume because they fill the tissues and maintain fluid volume more effectively than hypertonic or hypotonic solutions.
Why is it important for IV fluids to be isotonic?
It is important for IV to be isotonic to your blood so it doesn’t change the concentration of different molecules in the bloodstream. the cell wall will start to shrink, due to loss of the fluid. High osmotic pressure with allow fluids to pull fluids out of the cell.
How do isotonic solutions work?
If a cell is placed in an isotonic solution, there will be no net flow of water into or out of the cell, and the cell’s volume will remain stable. If the solute concentration outside the cell is the same as inside the cell, and the solutes cannot cross the membrane, then that solution is isotonic to the cell.
What are isotonic solutions and give one example?
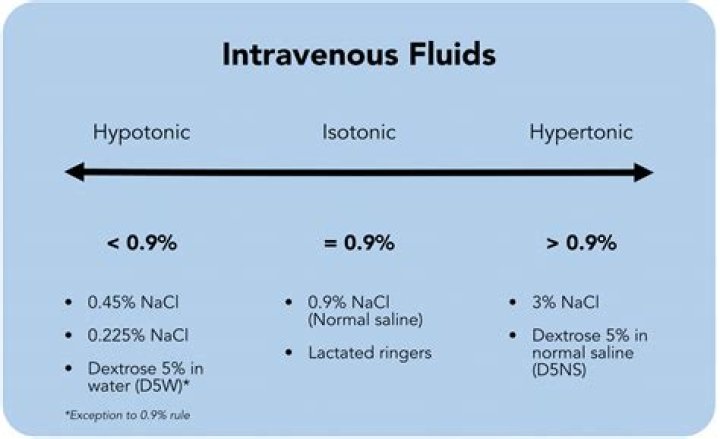
A solution is isotonic when its effective mole concentration is the same as that of another solution. This state provides the free movement of water across the membrane without changing the concentration of solutes on either side. Some examples of isotonic solutions are 0.9% normal saline and lactated ringers.
What is isotonic saline used for?
The fluid is called isotonic, as it does not change the size of the cells. Isotonic saline is used for ECF volume expansion, for irriga- tion, for correction of certain electrolyte disorders, and as a vehicle for i.v. administration of drugs.
Can I Nebulize with saline?
How to use Saline 0.9 % Solution For Nebulization. This medication is used with a special machine called a nebulizer that changes the solution to a fine mist that you inhale. Learn how to prepare the solution and use the nebulizer properly.
When would you use isotonic hypertonic and hypotonic solutions?
We give them an isotonic solution to try to expand the volume of their blood but we don’t want it to necessarily move solvent out of the vein into their tissues. Conversely the hypotonic solution is used when we need to put fluids into the cells for example if your patient is in Diabetic Ketoacidosis and HERE.
What IV fluids are isotonic?
Isotonic solutions are the third category of IV fluids. A solution is classified as isotonic when it has the same concentration of dissolved solutes as its outside environment. Biologically, isotonic solutions replace lost fluids while increasing blood vessel volume.
What is the difference between isotonic and hypotonic?
There are three classifications of tonicity that one solution can have relative to another.They are hypertonic, hypotonic, and isotonic. The key difference between Isotonic and Hypertonic is that the hypertonic solution contains more solvent than solute whereas the solute and solvent are equally distributed in the isotonic solution.
How to make isotonic saline solution?
How to Make Isotonic Saline Solution. Measure the amounts of water and salt needed for the solution. For each 8 oz. of tap water or filtered water, use 1/4 tsp. of salt. Use kosher, macrobiotic or table salt (NaCl).
Which is considered an isotonic solution?
A: An isotonic solution is made up of two or more substances and has the same concentration as the blood or cells in the body. An isotonic solution has a solvent inside as well as a solute. Isotonic solutions also have the same salt concentrations as the blood cells that surround them.



