What isomerism is butenedioic acid?
What isomerism is butenedioic acid?
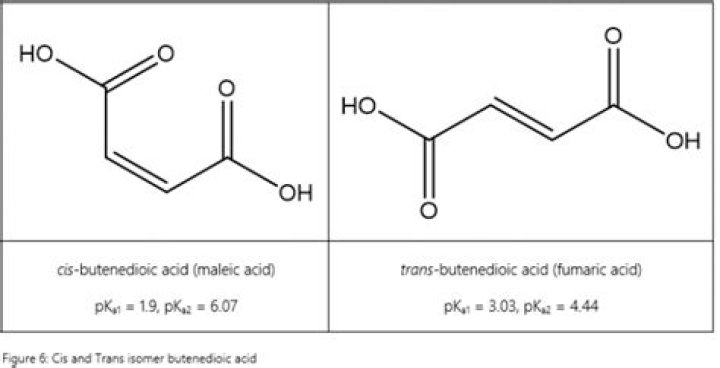
Maleic acid is the cis-isomer of butenedioic acid, whereas fumaric acid is the trans-isomer.
What isomerism is shown by fumaric acid?
Fumaric acid is the trans isomer of butenedioic acid, while maleic acid is the cis isomer.
What is the configuration of fumaric acid?
Fumaric acid
| PubChem CID | 444972 |
|---|---|
| Structure | Find Similar Structures |
| Chemical Safety | Laboratory Chemical Safety Summary (LCSS) Datasheet |
| Molecular Formula | C4H4O4 or COOH-CH=CHCOOH |
| Synonyms | fumaric acid 110-17-8 2-Butenedioic acid trans-Butenedioic acid Allomaleic acid More… |
What is the molecular formula for butenedioic acid?
C4H4O4
Fumaric acid/Formula
What is Z isomerism?
What are Z Isomers? Z isomers are alkenes having the substituents with higher priority on the same side of the double bond. The letter “Z” comes from zusammenin German, which means “together”.
What are the isomers of Cycloalkanes?
Like alkenes, cycloalkanes are capable of cis-trans isomerism. A cycloalkane has two distinct faces, and any substituent on a ring lies toward one of two faces. When two substituents on a ring point to the same face, they are cis. When the two substituents point to opposite faces, they are trans.
Why is E Z isomerism shown in some alkenes?
E−Z isomerism occurs because there is restricted rotation about double bonds.
What is Z in isomerism?
In Z isomers, the higher priority groups are on zee zame zide. In the letter E, the horizontal strokes are all on the same side; in the E isomer, the higher priority groups are on opposite sides. In the letter Z, the horizontal strokes are on opposite sides; in the Z isomer, the groups are on the same side.
What is lactic acid formula?
C3H6O3
Lactic acid/Formula
What is the proper formula for Bromous acid?
HBrO2
Answer: HBrO2 is the chemical formula of Bromous acid. Bromous acid is an inorganic compound with the formula HBrO2.
What is the pka1 of maleic acid?
The cis isomer is the less stable one of the two; the difference in heat of combustion is 22.7 kJ/mol. The physical properties of maleic acid are very different from that of fumaric acid….3D Structure for HMDB0000176 (Maleic acid)
| Property | Value | Reference |
|---|---|---|
| Melting Point | 130.5 °C | Not Available |



