What is the structure of Sulphur dichloride?
What is the structure of Sulphur dichloride?
Sulfur dichloride
| Names | |
|---|---|
| Structure | |
| Coordination geometry | C2v |
| Molecular shape | Bent |
| Hazards |
What is the formula for Disulfur?
Disulfur
| PubChem CID | 5460602 |
|---|---|
| Molecular Formula | S2 |
| Synonyms | disulfur sulfur dimer disulphur 23550-45-0 S2 More… |
| Molecular Weight | 64.13 |
| Dates | Modify 2021-12-05 Create 2004-09-16 |
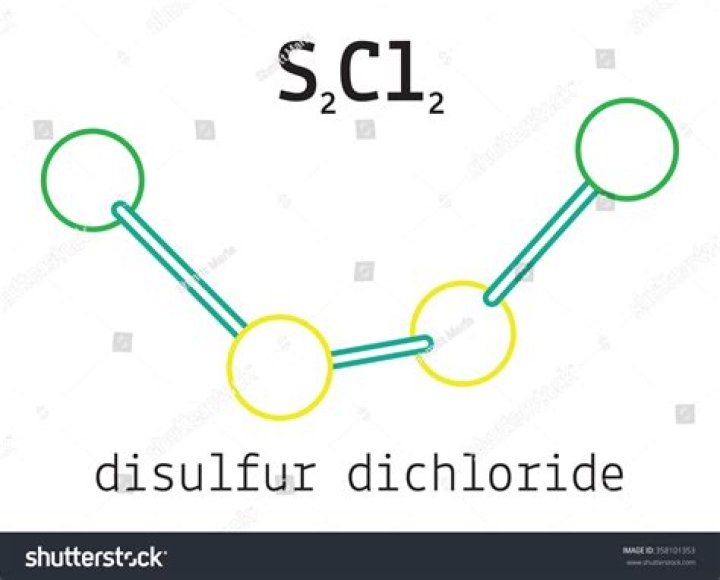
What is the covalent formula for Disulfur dichloride?
Cl2S2
Disulfur dichloride | Cl2S2 | ChemSpider.
What is the formula for Disulfur Difluoride?
F2S2
Disulfur difluoride/Formula
What is the correct formula for Disulfur pentoxide?
Disulfur dioxide, dimeric sulfur monoxide or SO dimer is an oxide of sulfur with the formula S2O2….Disulfur dioxide.
| Names | |
|---|---|
| Chemical formula | S2O2 |
| Molar mass | 96.1299 g/mol |
| Appearance | gas |
| Structure |
What is the formula for Disulfur trioxide?
Disulfur Trioxide S2O3 Molecular Weight — EndMemo.
How do you make Disulfur dichloride?
Synthesis, basic properties, reactions Pure disulfur dichloride is a yellow liquid that “smokes” in moist air due to reaction with water. An idealized (but complicated) equation is: 16 S2Cl2 + 16 H2O → 8 SO2 + 32 HCl + 3 S. It is produced by partial chlorination of elemental sulfur.
What is the chemical formula for Disulfur Decafluoride?
S2F10
Disulfur decafluoride/Formula
What is the formula of oxygen difluoride?
OF2
Oxygen difluoride/Formula
Is Disulfur dichloride ionic or covalent?
Sulfur dichloride is a covalent molecule.



