What is the shape of ch3nh2?
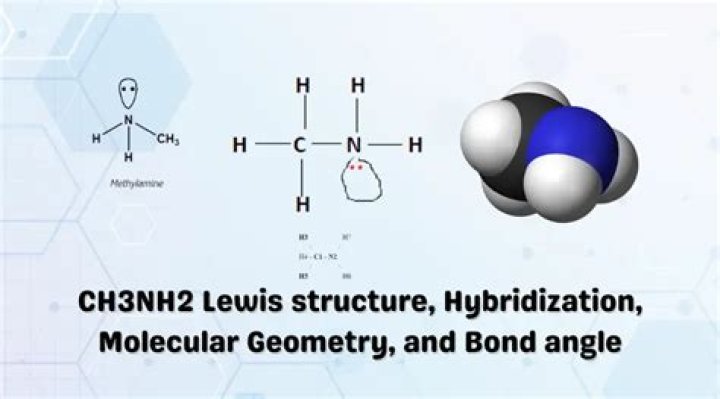
What is the shape of ch3nh2?
As a result, the methylamine molecule has a tetrahedral molecular shape considering carbon as a central atom.
How do you determine the polarity of a shape?
- If the arrangement is symmetrical and the arrows are of equal length, the molecule is nonpolar.
- If the arrows are of different lengths, and if they do not balance each other, the molecule is polar.
- If the arrangement is asymmetrical, the molecule is polar.
Is polarity affected by shape?
The shape of the molecule will determine the direction of each of the individual bond dipoles, and thus, will always play a role in determining the polarity of the molecule as a whole.
Are there polar bonds in CH3NH2?
Methyl amine ie CH3NH2 is a polar molecule. Polarity is the result of significant electronegativity difference between atoms.
What is the functional group of CH3NH2?
Question: Which functional group present in the compound CH3NH2? Answer: The functional group is primary amine . and the compund name is methylamine.
What shapes are polar and non-polar?
Lewis Structures and the Shapes of Molecules
| Formula | 3D Structure Shape Polarity | |
|---|---|---|
| 1. | CH4 | tetrahedral nonpolar |
| 2. | NH3 | trigonal pyramidal polar |
| 3. | H2O | bent polar |
| 4. | H3O+ | trigonal pyramidal charged |
Is CH3NH2 resonance structure?
The only way to draw any kind of resonance structure for this molecule would be to include some double-bonding from the lone pair of the Nitrogen to the Carbon, but this would give you pentavalent C, which is a no-no in Lewis Structures.
What factors affect the polarity of a molecule?
The polarity of a bond arises from the relative electronegativities of the elements. Electronegativity, it will be recalled, is the power of an atom of an element to attract electrons toward itself when it is part of a compound.
What factors affect the shape of a molecule?
The shape of a molecule is determined by the location of the nuclei and its electrons. The electrons and the nuclei settle into positions that minimize repulsion and maximize attraction. Thus, the molecule’s shape reflects its equilibrium state in which it has the lowest possible energy in the system.
What type of bond is CH3NH2?
CH3NH2 is able to form hydrogen bonds because hydrogen atoms are bound to a more electronegative atom, nitrogen.
What is the Lewis structure of CH3NH2?
In the CH3NH2 Lewis structure it makes sense to write the structure as written – a Carbon atom bonded to a Nitrogen atoms with Hydrogen atoms on the outside. The -NH2 part of CH3NH2 is called an amine functional group. In the Lewis structure for CH3NH2 there are a total of 14 valence electrons. This is the CH3NH2 Lewis structure: Methylamine.
What is the electronegativity of CH3NH2?
CH3NH2 is a Carbon has an electronegativity value of 2.5 , nitrogen’s electronegativity value is 3.0 and hydrogen has an electronegativity value of 2.2.
Is CH3NH2 polar or non polar?
Answer = CH3NH2 (Methylamine) is Polar. What is polar and non-polar? Polar. “In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole or multipole moment. Polar molecules must contain polar bonds due to a difference in electronegativity between the bonded atoms.
What is the molecular geometry of CH3NH2?
Re: Determining Molecular Geometry. In CH4, the molecular geometry is tetrahedral, so in C2H6 each C atom has a tetrahedral geometry In CH3NH2, the arrangement around C is tetrahedral but around N, the arrangement is pyramidal The -OH group in HCOOH has a bent arrangement as seen in water H-OH while the C atom has trigonal planar arrangement of H, O and OH.



