What is the molecular orbital electronic configuration of no?
What is the molecular orbital electronic configuration of no?
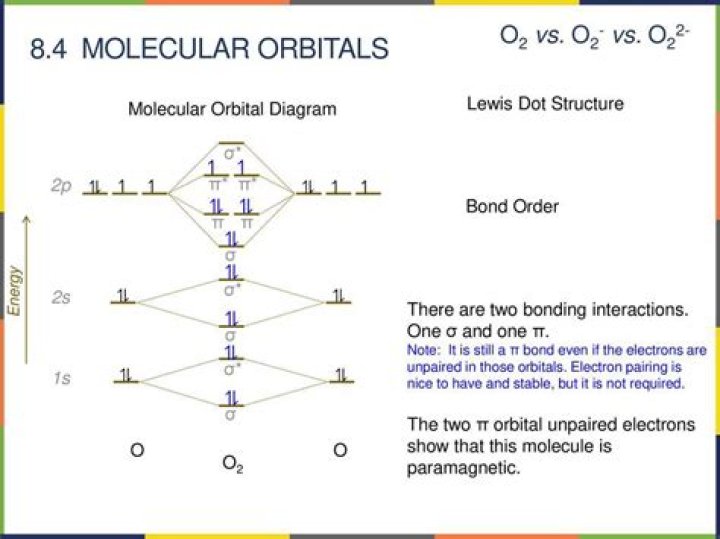
Because NO has 11 valence electrons, it is paramagnetic, with a single electron occupying the (π⋆2px,π⋆2py) pair of orbitals. Note that electronic structure studies show the ground state configuration of NO to be (σ2s)2(σ⋆2s)2(π2px,y)4(σ2pz)2(π2p⋆x,y)2 in order of increasing energy.
What is the bond order of NO+?
3
Bond order of NO+ is 3.
How do you find the molecular orbital electron configuration?
FUNDAMENTAL STEPS IN DERIVING MO DIAGRAMS
- Find the valence electron configuration of each atom in the molecule.
- Decide if the molecule is homonuclear of heteronuclear.
- Fill molecular orbitals using energy and bonding properties of the overlapping atomic orbitals.
- Use the diagram to predict properties of the molecule.
Are non bonding orbitals molecular orbitals?
A non-bonding orbital, also known as non-bonding molecular orbital (NBMO), is a molecular orbital whose occupation by electrons neither increases nor decreases the bond order between the involved atoms. Non-bonding orbitals are the equivalent in molecular orbital theory of the lone pairs in Lewis structures.
What is the electron configuration of mendelevium?
Rn 5f13 7s2
Mendelevium/Electron configuration
What is S and P mixing?
s-p mixing occurs when the s and p orbitals have similar energies. When a single p orbital contains a pair of electrons, the act of pairing the electrons raises the energy of the orbital. Thus the 2p orbitals for O, F, and Ne are higher in energy than the 2p orbitals for Li, Be, B, C, and N.
Why bond order of NO+ is 3?
– Higher the bond order of a bond, higher the stability of the bond. Hence can say that a single bond is weaker than a double bond and a double bond is weaker than a triple bond. – Here, \[N{{O}^{+}}\] has bond order equal to 3 that simply means that there is a triple bond present in between oxygen and nitrogen.
Why bond order of NO+ and N2 is same explain?
Bond order is the number of chemicalbondsbetween a pair of atoms. For example, in diatomic nitrogen N≡N thebond order is 3, in acetylene H−C≡C−H the bond order between the two carbon atoms is also 3, and the C−Hbond order is 1. NO+ has 10 electrons, a triple bond between N and O and two pairs, one on each atom.
What is the full form of BMO and ABMO?
Combination of two atomic orbitals results in formation of two molecular orbitals, bonding molecular orbital (BMO) whereas other is anti-bonding molecular orbital (ABMO).
What is the electron configuration of fermium?
Rn 5f12 7s2
Fermium/Electron configuration



