What is the electron of rhenium?
What is the electron of rhenium?
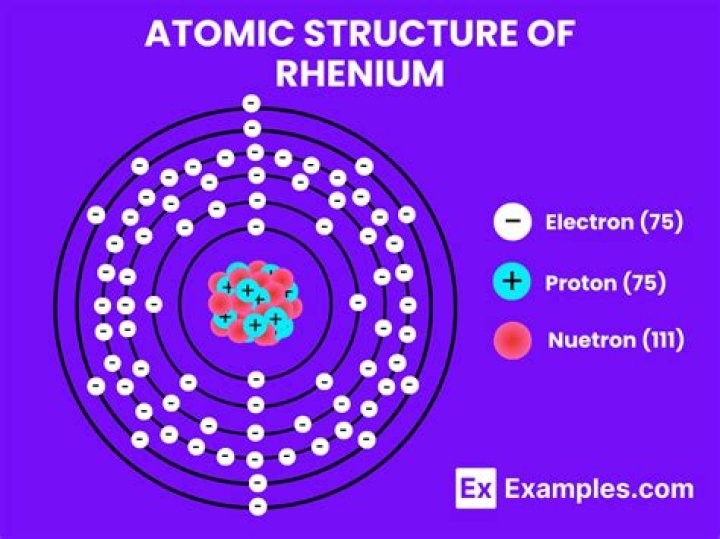
75 electrons
Rhenium atoms have 75 electrons and the shell structure is 2.8. 18.32. 13.2.
How many electrons does rhenium have?
2, 8, 18, 32, 13, 2
Rhenium/Electrons per shell
What is Argons electron?
So… for the element of ARGON, you already know that the atomic number tells you the number of electrons. That means there are 18 electrons in an argon atom.
How many valence electrons are in re?
Rhenium contains 2 electrons in its outer shell, thus it has 2 valence electrons.
What is re on the periodic table?
rhenium (Re), chemical element, a very rare metal of Group 7 (VIIb) of the periodic table and one of the densest elements.
What does re mean on the periodic table?
Rhenium is a chemical element with the symbol Re and atomic number 75. It is a silvery-gray, heavy, third-row transition metal in group 7 of the periodic table. With an estimated average concentration of 1 part per billion (ppb), rhenium is one of the rarest elements in the Earth’s crust.
What is Re on the periodic table?
What does Re mean on the periodic table?
What is Argons number of neutrons?
22 neutrons
There are 22 neutrons in one atom of argon. On the periodic table, argon’s atomic number is 18. This tells you that it has 18 protons.
What is meant by 3p3?
#33 what is meant by 3p3? 3rd orbital in p sublevel 3 electrons. #34a give the electron configuration for Na. 1s2 2s2 2p6 3s1.
What does re stand for on the periodic table?
How many protons does RE have?
75
Rhenium/Atomic number
Rhenium (Re). Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of rhenium-186 (atomic number: 75), an isotope of this element. The nucleus consists of 75 protons (red) and 111 neutrons (orange).
What is the electron configuration of rhenium 75?
Rhenium is a chemical element with atomic number 75 which means there are 75 protons and 75 electrons in the atomic structure. The chemical symbol for Rhenium is Re. Electron configuration of Rhenium is [Xe] 4f14 5d5 6s2. Possible oxidation states are +4,67.
What is the chemical symbol for rhenium?
The chemical symbol for Rhenium is Re. Electron configuration of Rhenium is [Xe] 4f14 5d5 6s2. Possible oxidation states are +4,67. The periodic table is a tabular display of the chemical elements organized on the basis of their atomic numbers, electron configurations, and chemical properties.
Is rhenium a solid at room temperature?
Rhenium is a chemical element with symbol Re and atomic number 75. Classified as a transition metal, Rhenium is a solid at room temperature. J.C. Slater, J Chem Phys, 1964, 41 (10), 3199-3205.
What are the properties of rhenium superconductors?
Rhenium metal superconducts at 1.697 ± 0.006 K. In bulk form and at room temperature and atmospheric pressure, the element resists alkalis, sulfuric acid, hydrochloric acid, dilute (but not concentrated) nitric acid, and aqua regia .



