What are the examples of dative bond?
What are the examples of dative bond?
An example of a dative covalent bond is provided by the interaction between a molecule of ammonia, a Lewis base with a lone pair of electrons on the nitrogen atom, and boron trifluoride, a Lewis acid by virtue of the boron atom having an incomplete octet of electrons.
Does bcl3 have a dative bond?
B3N3H6
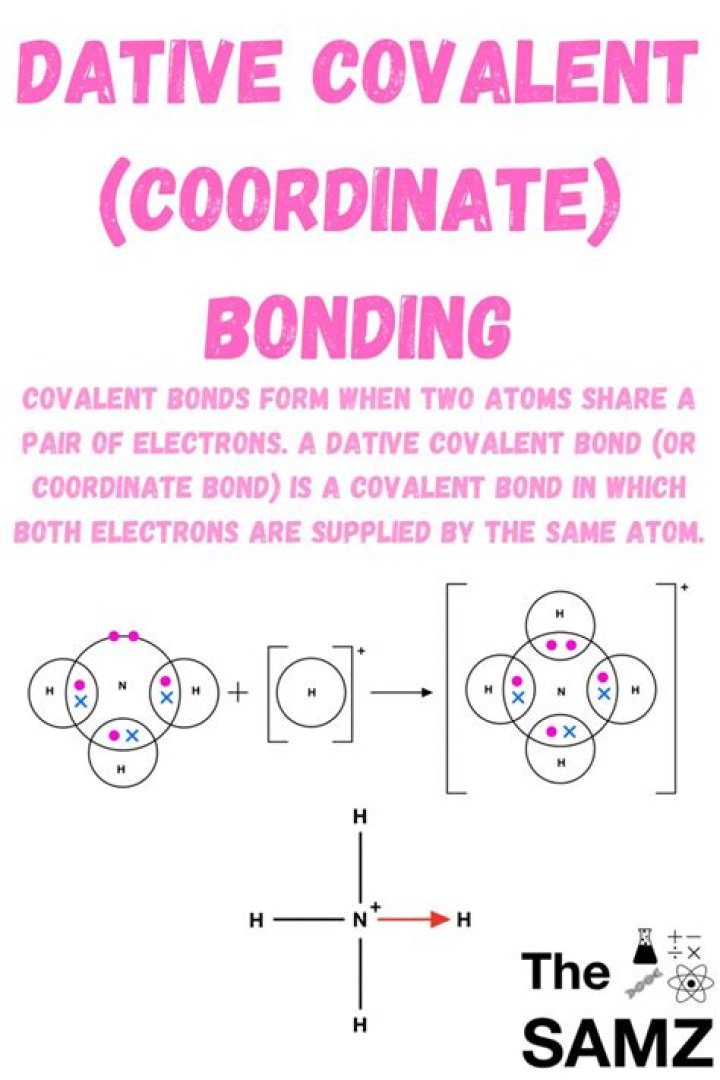
What is dative bond explain?
A dative bond is a covalent bond between two atoms where one of the atoms provides both electrons that form the bond. A dative bond is also known as a dipolar bond or coordinate bond.
What is dative pi bond?
A coordinate bond (also called a dative covalent bond) is a covalent bond (a shared pair of electrons) in which both electrons come from the same atom. A covalent bond is formed by two atoms sharing a pair of electrons. The atoms are held together because the electron pair is attracted by both of the nuclei.
What bond is CaF2?
ionic bond
This is how the ionic bond forms in Calcium Fluoride (CaF2).
In which of the following the dative bond is not present?
Here in this question we have tried to complete the octet of atoms in the compounds by giving them correct electrons and then we get that only hydrazine does not have dative bonding.
Why does SO3 have dative bonds?
The molecule $S{O_3}$ has two coordinate bonds but that structure is not the most stable form as it carries a formal charge. – In the molecule $N{H_3}$ the structure has a lone pair of electrons present on its central nitrogen atom which can give it to form a dative bond.
Does N2O contain dative bond?
Dative bond is not present in 1) BF3 2) N2O.
How many dative bonds does SO3?
The molecule $S{O_3}$ has two coordinate bonds but that structure is not the most stable form as it carries a formal charge.
Is dative bond PI or Sigma?
Sigma bonds are always single bonds. One electron is usually provided by each of the overlapping orbitals, although in certain cases both of the electrons may be donated by the same atom. The second case is called dative covalent bonding. Pi bonds can only form AFTER sigma bonds have already formed.
Which hydrogen bond is strongest?
Models of Strong Hydrogen Bonds The strongest OHO bonds are in ionic systems, since electrostatic attraction between a dipole and a monopole is generally greater than between two dipoles. Also, the greater electrostatic attraction leads to a shorter dOO, which correlates with the greater strength.
What type of bond is I and CL?
polar covalent bond
A polar covalent bond exists when atoms with different electronegativities share electrons in a covalent bond. Consider the hydrogen chloride (HCl) molecule….Polar Covalent Bonds.
| Structural Unit1 | Bond Moments (D) |
|---|---|
| C—Cl | 1.5 |
| C—Br | 1.4 |
| C—I | 1.2 |
| C = O | 2.3 |
When to use the adverb anhand?
This use is mandatory when the genitive case in the plural number is indiscernible or when the noun stands alone in the singular number. Note : anhand can also be used as an adverb when combined with the preposition “von”.
Is Mithilfe dative or dative case?
When the genitive case in the plural number is indiscernible, the noun must be in the dative case. Nouns that stand alone in the singular number are usually not inflected. Note : mithilfe can be used as an adverb, combined with the preposition “von” when the noun has no article.
When do you use dative in German?
When the genitive case in the plural number is indiscernible, the use of dative is mandatory. Nouns that are in the singular number are usually not inflected. Note : unterhalb can also be used as an adverb, when combined with the preposition “von”, mainly with place names and numbers without an article.
What is the difference between Laut and dative case?
Note : laut also governs the dative case. When the genitive case in the plural number is indiscernible, the noun must be in the dative case. Nouns that stand alone in the singular number are usually not inflected. Note : mithilfe can be used as an adverb, combined with the preposition “von” when the noun has no article.



