How the naphtha is reformed in a catalytic reformer?
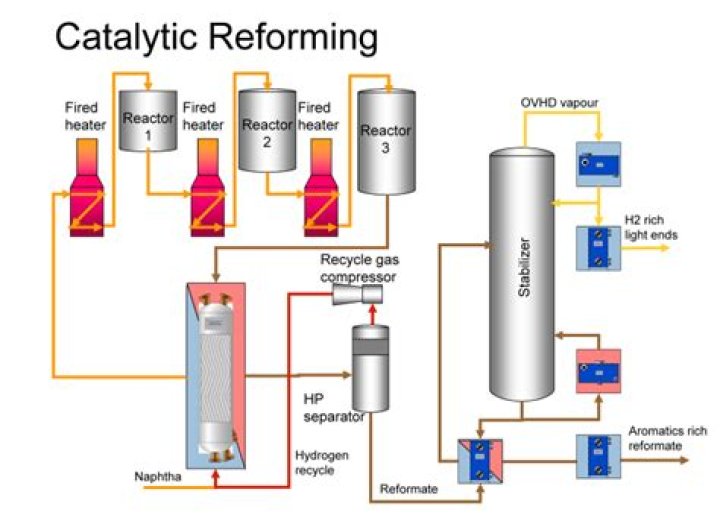
How the naphtha is reformed in a catalytic reformer?
In a typical reforming unit the naphtha charge is first passed over a catalyst bed in the presence of hydrogen to remove any sulfur impurities. The desulfurized feed is then mixed with hydrogen (about five molecules of hydrogen to one of hydrocarbon) and heated to a temperature of 500–540 °C (930–1,000 °F).
Which catalyst is used in naphtha reforming?
Naphtha reforming catalyst is a bifunctional catalyst consists of a metal function, mainly platinum, and an acid function, usually chloride alumina. The metal function catalyzes the hydrogenation and dehydrogenation reactions and the acid function promote the isomerization and cyclization reactions [11], [12], [13].
What is CCR unit?
Continuous Catalytic reforming (CCR) is a chemical process that converts petroleum refinery naphthas distilled from low-octane oil into high-octane liquid products called reformates, which are premium blending stocks for high-octane gasoline.
What is CCR unit in refinery?
Central to these process units and streams is the Catalytic Reformer (CCR) unit. This unit takes low-value heavy naphtha from the CDU and converts it, after hydro-treating, into a higher- value high-aromatics, high-octane feedstock. Note: in this paper, CCR is used as a generic to indicate a catalytic reforming unit.
Is catalytic reforming exothermic or endothermic?
Catalytic steam reforming reaction produces carbon monoxide and hydrogen from methane and water. This reaction is a large endothermic reaction. Industrial hydrogen production is mainly produced by this reaction. This reaction is also used for hydrogen, which is a feedstock of methanol [95].
What is the difference between isomerization and reforming?
Catalytic reforming is the process of transforming C7–C10 hydrocarbons with low octane numbers to aromatics and iso-paraffins which have high octane numbers. It is a highly endothermic process requiring large amounts of energy. Isomerization is a mildly exothermic reaction and leads to the increase of an octane number.
How does CCR reactor work?
Continuous catalyst regeneration (CCR) is part of the catalytic reforming process in a refinery where hydrogen and naphtha feed are reacted to create desired end products. The catalyst used in the reaction is continuously flowing through the reactors and then regenerated.
What is the hydrocracking process?
Hydrocracking is a process to convert larger hydrocarbon molecules into smaller molecules under high hydrogen pressure and elevated temperature. It is commonly applied to upgrade the heavier fractions of the crude oils to produce higher value transportation fuels.
What is the difference between reforming and isomerization naphtha?
How does catalytic reforming work?
Catalytic reforming uses a catalyst, usually platinum, to produce a similar result. Mixed with hydrogen, naphtha is heated and passed over pellets of catalyst in a series of reactors, under high pressure, producing high-octane gasoline.
What is the difference between reforming and isomerization Naphtha?
What is the reforming process?
reforming, in chemistry, processing technique by which the molecular structure of a hydrocarbon is rearranged to alter its properties. The process is frequently applied to low-quality gasoline stocks to improve their combustion characteristics.
How does temperature affect naphtha component conversion?
The optimization results indicated that heavy naphtha component conversion (paraffin’s and naphthenes) increases with an increasing in reaction temperature and pressure while decreases with an increase in LHSV. The kinetic study of catalytic reforming reactions reported helped establishing the reaction model explicitly.
How is naphtha transformed into reformates?
Naphtha is transformed into reformates through catalytic reforming process which requires the reconstruction of low-octane hydrocarbons in the petroleum naphtha into more valuable high octane gasoline components. Naphtha and reformate are complex mixtures of paraffin’s, naphthenes, and aromatic in the C5–C12 range of naphtha compositions.
How is naphtha converted into aromatic gasolines?
Catalytic naphtha reforming is the main process producing aromatic gasolines. “Octanizing” catalytic reforming processes are carried out in oil refineries to convert desulfurized and usually dehexanized straight-run naphtha into high-octane number aromatic-rich gasoline (the reformate,.
What is the temperature of thermoplastic reform?
Thermal reforming employed temperatures of 510–565 °C (950–1,050 °F) at moderate pressures—about 40 bars (4 MPa), or 600 psi—to obtain gasolines (petrols) with octane numbers of 70 to 80 from heavy naphthas with octane numbers of less than 40. The product yield, although of a higher octane level, included olefins, diolefins, and aromatic compounds.